Technology highlights – Photo-acoustic Imaging (PAI)
Posted by Johanna Bischof, on 11 November 2020
Interview with Enzo Terreno, Ph.D. from the University of Torino, Italy.
Please tell us a bit about yourself and the facility where you work.
My name is Enzo Terreno, and I have the pleasure to be the Coordinator of the Multi-Modal Molecular Imaging (MMMI) Italian Node belonging to Euro-BioImaging. This is a multi-sited Node comprising 8 imaging centers distributed across 4 cities (Torino, Pisa, Milano, and Napoli). As the central coordinator, I’m based in Torino.

We are today talking about Photo-acoustic Imaging (PAI). Can you please tell us how PAI works?
Photoacoustic imaging is an emerging and powerful technology with a wide spectrum of applications in the field of in vivo imaging. The special mechanism of contrast generation, combined with impressive recent technical developments, has significantly boosted the performance, and extended the application limits of the technique. PAI combines the high-contrast and the spectroscopic features of optical imaging in the near-infrared region (NIR) with the high spatial resolution and better tissue penetration of ultrasound imaging.

Briefly, PAI contrast originates from the absorption of a pulsed laser beam (typically in the range of 600 – 1000 nm) by a chromophore that causes a transient thermoelastic expansion of the surrounding tissues, responsible for the generation of acoustic waves that are detected by a broadband US transducer and converted into images.
That’s why PAI is considered the “sound of the light”.
The investigation range of in vivo PAI spans from a few millimeters to centimeters of depth with an excellent spatial resolution of a few hundreds of microns, attainable with and without using contrast agents. Though better than pure optical imaging techniques, the penetration depth of PAI does not allow for the whole-body detection that is possible with CT, MRI, and nuclear medicine technologies. Therefore, clinical applications are limited to the analysis of superficial organs and structures (e.g. palmar vessels detection, breast analysis).
The resolution (for tomographic application) is also slightly lower than US, MRI and CT, but definitively better than PET/SPECT and pure optical imaging. The sensitivity detection of PAI is in the middle between highly sensitive nuclear medicine, and pure optical technologies and less sensitive MRI, US, and CT.
Finally, it is worth remembering that PAI does not use ionizing radiations and can therefore be considered a safe technology.
What are some possible applications of PAI?
At the preclinical level, the spectrum of the possible applications of PAI coregistered with micro-US at high resolution is very broad, ranging from oncology (cancer detection, assessment of vascular volume and hypoxia, targeting experiments, assessment of drug distribution and therapy outcome), cardiovascular biology (O2 saturation and hypoxia), neurobiology (stroke, brain cancer, intracranial injection of drugs, functional imaging), development biology (vascular volume and oxygenation of placenta, image-guided embryo injection, fetal analysis), abdomen analysis (kidney diseases as renal microcirculation flow, renal obstruction analysis, gastrointestinal motility, organs’ perfusion), skin analysis (mainly detection of melanin in melanoma cells), to image-guided injection of drugs.
In principle, similar applications could be translated into clinical settings, though, as pointed out above, the studies are limited to superficial tissues.
Although the development of specific probes for PAI detection is a very active and productive research area, most of the above-mentioned applications exploit endogenous PAI probes, like hemoglobin (oxy- and de-oxy-Hb have distinct signals, thus allowing the monitoring of tissue oxygenation), melanin, and recently, also lipids and water, thus significantly favoring the clinical translation of the technology.
What kinds of research are your facilities involved it that use PAI? What scientific questions are you addressing?
The (preclinical) research activity on PAI at MMMI Italian Node is mainly focused on addressing the following topics:
- Exploit endogenous PAI contrast to assess vascular volume and oxygenation levels in tumours
- Design and validate innovative PAI probes, including systems for multimodal detection
- Physiopathological studies (O2-saturation, hypoxia)
- Pharmacokinetic for studying the distribution of labelled molecules of nanostructured materials.
Because of the wide variety of research, we do using PAI, I’ll very briefly tell you two examples:
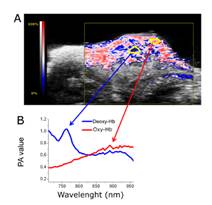
PAI was used in the study on MRI Method To Map Tumor Hypoxia Using Red Blood Cells Loaded with a pO2-Responsive Gd-Agent [1]. Here PAI allowed researchers to non-invasively assess tumor vascular volume and tumor hypoxia, without the use of exogenous contrast agents. This allowed them to monitor tumor growth and aggressiveness and validated MRI results on vascular volume and hypoxia.
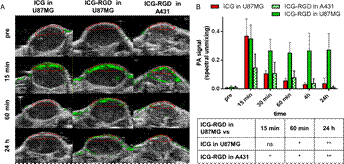
In this study, Photoacoustic imaging of integrin-overexpressing tumors using a novel ICG-based contrast agent in mice [2], a novel integrin-targeting photoacoustic probe was synthesized to combine the good PAI properties of ICG and the favourable αvβ3-binding capabilities of a cyclic RGD peptidomimetic moiety previously used for fluorescence imaging.
What are some challenges of this kind of imaging? What do researchers have to pay attention to when performing these experiments?
The challenges for the experimentation are related to the two main limitations of the technology:
1) The intrinsic background PA signal from some endogenous chromophores (mainly melanin and hemoglobin) that can limit the detection sensitivity, especially for the detection of exogenous probes (e.g. ICG). Furthermore, melanin requires the removal of hairs and the use of black (or colored) animal strains.
2) The limited penetration depth that makes possible whole-body experiments only on small animals (e.g. mice), whereas only superficial organs can be visualized in larger animals.
What other services do you provide in your facility that would be useful in combination with this type of imaging?
The MMMI Italian Node is equipped with all the state-of-the-art in vivo imaging technologies. Among them, high frequency ultrasound is naturally the technique most commonly combined with PAI, and all the PAI scanners in the Node are fully integrated with Ultrasound imager. In principle, PAI can be combined with optical and MR scanners, though efforts are required for allowing the co-localization of the two images.
Why should scientists choose your facility for using this technology?
The three Centers of the MMMI Italian Node equipped with PAI technology – Department of Molecular Biotechnology and Health Science in Torino, Clinical Physiology Institution CNR in Pisa, and Biostructures and Bioimages Institute CNR in Napoli – have also a strong competence and active collaborations for the design of innovative PA probes. Furthermore, the MMMI Italian Node has a longstanding and strong expertise in all the molecular and clinical imaging technologies represented in Euro-BioImaging (MRI, nuclear medicine, optical imaging, ultrasound), thus allowing the execution of validation and comparative investigations on site.
Want to use Photo-acoustic Imaging via the Euro-BioImaging service?
Euro-BioImaging is the European landmark research infrastructure for biological and biomedical imaging as recognised by the European Strategy Forum on Research Infrastructures (ESFRI). All scientists, regardless of their affiliation, area of expertise or field of activity can benefit from Euro-BioImaging’s pan-European open access services. By facilitating user access to high quality imaging facilities, resources, and services, with a constantly evolving technology offer, Euro-BioImaging will boost the productivity and impact of research across Europe.
Potential users of the Euro-BioImaging technologies are encouraged to submit project proposals via our website. To do so, you can LogIn to access our application platform, choose the technology you want to use and the facility you wish to visit, then submit your proposal. By using Euro-BioImaging, users will benefit from advice and guidance by technical experts working at the Nodes, training opportunities, and data management services.
For more information visit our website at www.eurobioimaging.eu or contact us at info@eurobioimaging.eu


Enzo Terreno
University of Torino
Italy


 (No Ratings Yet)
(No Ratings Yet)
Hello .first time in 2017 in Amesterdam in phylips center heard the future of diagnosis of C A D is ultrasound not the x ray so it was exciting for me to think and learn and use this technology in my field