OpenFlexure: an open-source 3D printed microscope
Posted by Joe Knapper, on 19 October 2021
The OpenFlexure Microscope is an open-source microscope 3D printed in plastic, with versions suitable for hobbyists, researchers, and lab technicians. Currently used around the world as an accessible, customisable alternative to expensive and fixed commercial microscopes, the OpenFlexure Microscope began life as an academic prototype for holding a lens and sample in place. Here we discuss how the project has developed, with applications including malaria diagnosis, super-resolution imaging and remote fieldwork. The project has also expanded as a community, with an online forum, accessible software and an international network of users.

The overarching aim of the OpenFlexure project is “Microscopy for Everyone”. Traditionally an area with a high barrier to entry, microscopy is more accessible than ever thanks to a recent surge in the availability of 3D printing and open-source knowledge sharing. The OpenFlexure Microscope (Fig 1) lowers the cost of a research grade microscope by two orders of magnitude compared to commercial alternatives. A lab grade OpenFlexure Microscope features a Raspberry Pi computer controlling a camera and translation stage, allowing users to leave the microscope to capture high-magnification images over a large area or extended time period [1]. First described in a 2016 paper [2], the OpenFlexure Microscope has since been used in over a dozen countries worldwide, and has been adapted for a range of imaging modes, budgets, and users, ranging from students and hobbyists to researchers and healthcare professionals.

Shown in Fig 2, the first design of the OpenFlexure Microscope was considerably less versatile. It was designed to replace the costly and heavy structure of a commercial, metal microscope with a low-cost, 3D printed mechanism. It held a lens and sample in place, allowing the user to adjust the focus by moving the lens relative to the sample. Rather than attempting to replicate the dovetail slide used in metal microscopes to facilitate movement, the design instead capitalised on the flexibility of plastic. By applying a force, the plastic could be flexed and bent, moving the lens and adjusting the focus. As the microscope gained features in the subsequent years, the flexure hinges remained, and give the OpenFlexure Microscope its name.
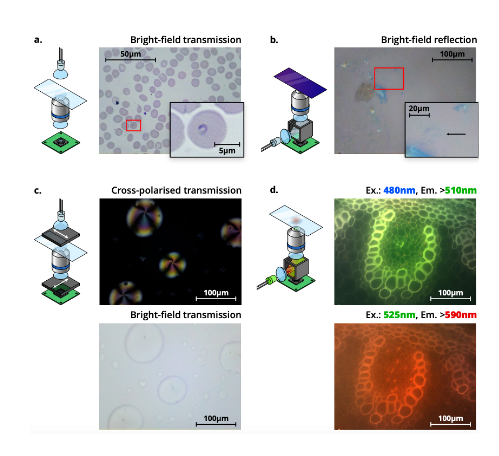
These hinges were used in later versions to increase the degrees of freedom in positioning from one to three; allowing the user to position a sample in x and y, then focus by moving the optics in z. Rather than positioning by hand, a Raspberry Pi, motor controller board and stepper motors gave users the option to automate the positioning and focus. Support for higher power magnification by including standard RMS objectives increased the number of potential applications, as did mounts for dedicated LED illumination. Rather than relying on users directly observing samples, the inclusion of a webcam or Raspberry Pi Camera allowed images to be captured, stored, and shared. The OpenFlexure Microscope has also been developed to support imaging modes including brightfield transmission, darkfield transmission, reflection, fluorescence, and cross-polarised transmission (Fig 3).
Today the OpenFlexure Microscope has multiple variants, and two spin-off projects. The Delta Stage, a larger microscope which moves the sample in x, y, z, allowing larger optics to be held static, and the Block Stage, a high precision 3-axis translations stage that can be used to automatically align optical fibre (both shown in Fig 4). Low-cost versions of the OpenFlexure Microscope can be printed and assembled for less than $20, while a fully automated research grade version costs around $200. Software is included to perform movements, autofocus, captures and automated scanning. From super-resolution research in labs in Glasgow to remote fieldwork in Panama and the Antarctic, researchers have used, developed, and customised the microscope for a range of uses and environments.

The main focus of OpenFlexure Microscope development today is for use in Tanzania. In areas of sub-Saharan Africa, malaria is still endemic and kills over 250,000 people a year. Although serious, malaria is treatable and the prognosis generally positive if diagnosed early. The World Health Organisation describes manual optical microscopy as the “gold standard” of malaria diagnosis – staining and imaging a blood sample to search for the plasmodium parasites that cause the disease. Requiring expensive, heavy, and immobile commercial microscopes, this technique is unavailable in many of the areas most affected by malaria.
This is where the low cost, accessibility and distributed manufacturing of the OpenFlexure Microscope has clear benefits over traditional microscopy. Using a standard 100× RMS objective and LED illumination, the disease-causing protozoa can be resolved, with images saved for future reference and training. Manufactured by our partners at BongoTech and used by the Ifakara Health Institute in Tanzania, the microscope is being tested for use in remote health clinics. This application has taken OpenFlexure Microscope development in a new direction; rather than focusing on adding features, the emphasis is on verifying and certifying every relevant component. For the microscope to become the first in-vitro diagnostic medical device made in Tanzania, the manufacturing, use and maintenance of the microscope must be understood by all parties. The technical challenges of this co-development between the UK and Tanzania are detailed by Stirling et al. [3]
As work is ongoing to certify the essential medical components of the OpenFlexure Microscope, developers and the community continue to add new features for researchers and hobbyists. The launch of a forum allows users to share suggestions, requests and help with the community [4]. The OpenFlexure software allows users to calibrate, control and automate a network of microscopes. The open-source nature of the project allows unprecedented accessibility to high performance microscopy, removing barriers to entry and working towards the project goal of “microscopy for everyone”.
The OpenFlexure Microscope source files, assembly instructions and other relevant links are all freely available at https://openflexure.org/.
References
[1] Joel T. Collins et al., “Robotic microscopy for everyone: the OpenFlexure microscope” Biomed. Opt. Express 11, 2447-2460 (2020); https://doi.org/10.1364/BOE.385729
[2] James P. Sharkey et al., “A one-piece 3D printed flexure translation stage for open-source microscopy” Review of Scientific Instruments 87, 025104 (2016); https://doi.org/10.1063/1.4941068
[3] Julian Stirling et al., “The OpenFlexure Project. The technical challenges of Co-Developing a microscope in the UK and Tanzania” 2020 IEEE Global Humanitarian Technology Conference (GHTC), (2020); https://doi.org/10.1109/GHTC46280.2020.9342860


 (No Ratings Yet)
(No Ratings Yet)