Time-resolved confocal fluorescence microscopy from PicoQuant
Sponsored by PicoQuant, on 31 January 2024
On January 11th Professor Samrat Mukhopadhyay shared insights into the complicated behavior of the FUS low-complexity domain during liquid-liquid phase separation. He and his team study protein dynamics at the single molecule level with Fluorescence Correlation Spectroscopy (FCS), single-molecule FRET (smFRET), and anisotropy to gain a comprehensive understanding.
His excellent presentation sparked great interest:
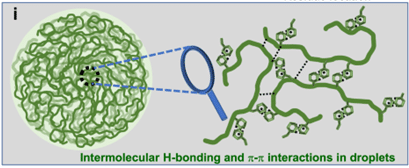
- about the molecular mechanisms that underlie the formation of condensates,
- how a protein’s amino acid sequence, particularly in intrinsically disordered regions (IDR), determines phase separation behavior,
- how the presence of multiple components inside cells may give specificity to condensate formation
All these questions are active areas of research. Furthermore, lively in-depth discussions of different FRET implementations started:
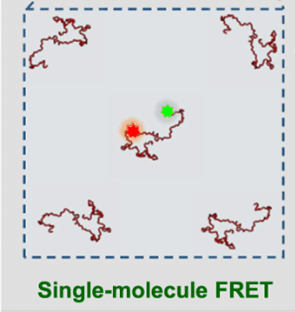
- Single-molecule FRET measurements in single condensates
- possible intermolecular FRET in dense condensates in addition to intramolecular FRET
- FRET imaging of condensates with acceptor photobleaching
- How to investigate protein complex formation with FRET
They illustrated once more that the FRET effect is very versatile and if interpreted correctly can provide valuable insights.
You can find the webinar recording here: https://focalplane.biologists.com/2024/01/31/focalplane-features-samrat-mukhopadhyay-with-picoquant/
For his experiments, Professor Mukhopadhyay used a time-resolved confocal fluorescence microscope from PicoQuant, the MicroTime 200. The microscope’s modular configuration can be flexibly adapted so that all the above-mentioned techniques can be applied to the same sample with the same instrument:

For smFRET experiments, he employed two excitation lasers for pulsed interleaved excitation, which controls for the presence of the acceptor by directly exciting it. The fluorescence was split onto two detectors for recording donor and acceptor emission. From the ratio of both signals, the FRET efficiency and stoichiometry per burst are calculated.
For FCS, it is also beneficial to split the signal onto two detectors and to cross-correlate them. This eliminates detector afterpulsing, which is a common artifact. FCS reports on the concentration and diffusion behavior of labeled species, while FRET reports on conformational states and their changes.
For time-resolved anisotropy and anisotropy imaging, the fluorescence was separated with a polarizing beam splitter into parallel and perpendicular channel. The ratio of both signals reveals the molecule’s orientation. By fitting time-resolved anisotropy curves, one can retrieve the rotation time or chain reorientation time of the molecule.
You can check all the details here:
Joshi, A., Walimbe, A., Avni, A. et al. Single-molecule FRET unmasks structural subpopulations and crucial molecular events during FUS low-complexity domain phase separation. Nat Commun 14, 7331 (2023). https://doi.org/10.1038/s41467-023-43225-y
Building on two decades of experience with the MicroTime 200, PicoQuant recently launched a new single photon counting confocal microscope, Luminosa. It also features smFRET, FCS, and anisotropy, among other techniques. The system software offers a context-based workflows for each method, which increases ease of use and saves time. It also includes online previews of the data during acquisition, so that users can quickly check sample and data quality and adjust parameters right away.
The enhanced integration of hardware and software in Luminosa enables many new features that increase both ease of use and reproducibility:
- The sample-free auto-alignment ensures that you always measure at optimal conditions with the highest sensitivity.
- The excitation laser power is automatically calibrated and can be set in µW, so that you always measure with the same conditions.
- The configuration of hardware, software interface, and analysis channels can be saved and loaded again automatically. This saves valuable time, avoids accidental errors, increases reproducibility of experiments, and moreover allows you to quickly switch between techniques to investigate the sample.
- The variable PSF feature enables you to switch between diffraction limited and larger observation volume for smFRET and FCS measurements, to match the observation time to the molecular dynamics and diffusion speed.
Learn more about the capabilities of Luminosa: https://www.picoquant.com/products/category/fluorescence-microscopes/luminosa-single-photon-counting-confocal-microscope#custom2
To read about investigations of synapsin-1 liquid-liquid phase separation with Luminosa using FLIM and multi-point FCS, check the application note here: https://www.picoquant.com/phase-separation



 (No Ratings Yet)
(No Ratings Yet)