Spider-like neurons finding their way: signposts for multipolar migration in the developing brain
Posted by Jaakko Lehtimäki, on 11 March 2026
Developing neurons travel large distances and orchestrate into precise layers to enable functional wiring of the brain. Within this unique tissue environment, many neurons use their neighbors like railway tracks, navigating in a directed manner toward their correct layer. However, this is not always the case. Multipolar migration, where cells extend multiple protrusions and display seemingly unpredictable movement patterns, is used by most developing neurons and observed across vertebrates (Noctor et al., 2004; Tabata & Nakajima, 2003). As mainly descriptive models existed for how and why this wandering type of migration occurs, we explored how neurons undergoing multipolar migration navigate within the part of the brain responsible for transmitting visual information – the retina (Lehtimäki et al., 2026).
Zebrafish retina: a window to study multipolar neuronal migration
In my PhD, I studied the intracellular force-generation mechanisms of migrating cells on a dish and gradually grew frustrated with how artificial that environment was for the cells. For my postdoc, I therefore joined the Norden lab to learn how to probe cell migration mechanics in vivo in zebrafish, a vertebrate model with unmatched live-imaging potential. I was intrigued by neuronal migration in the developing brain, which largely lacks prominent fibrous extracellular matrix (ECM) that many other cell types in the body use for translocation (Ruoslahti, 1996). Further, seeing spider-like multipolar neurons on the move for the first time (Figure 1B), made me dive into finding how do they move to their correct location within this unique tissue milieu.
We used developing zebrafish retina as an evolutionary conserved and optically transparent part of the brain (Hahn et al., 2023), to study multipolar neuronal migration. The fast development of zebrafish allowed us to document the entire retinal layering process in a single 48-hour live-imaging session on a spinning disk confocal. I focused on retinal interneurons called horizontal cells (HC), which use multipolar migration to travel across the developing retina and reach the photoreceptors. There, HCs later trim visual information before it is transmitted to the brain (Figure 1A). Previous work from the lab had shown that HCs display this wandering-type migration in an environment free of fibrous ECM (Amini et al., 2022). This raised an interesting question: could these cells rely on extrinsic factors (contact-mediated or secreted) to successfully navigate?
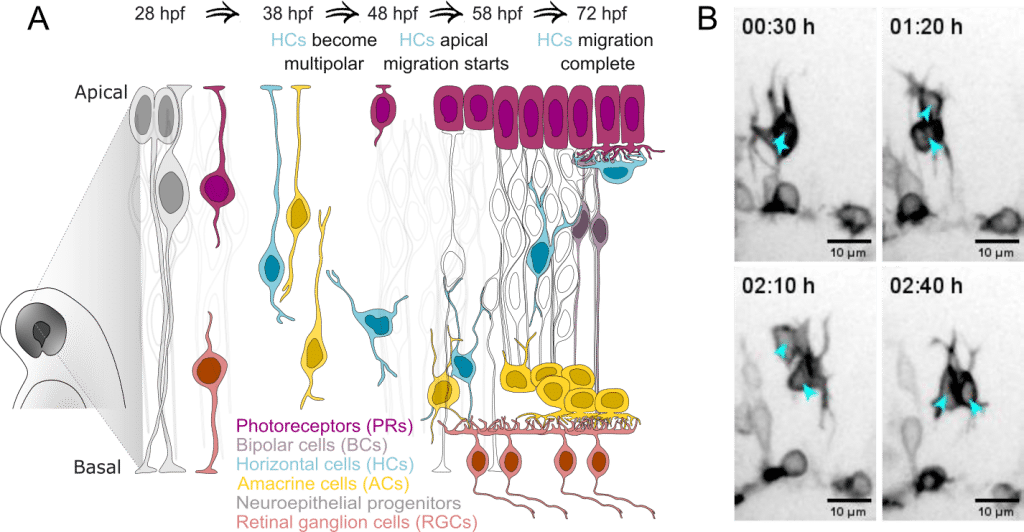
Migrating horizontal cells ignore most neighbors, except amacrine cells.
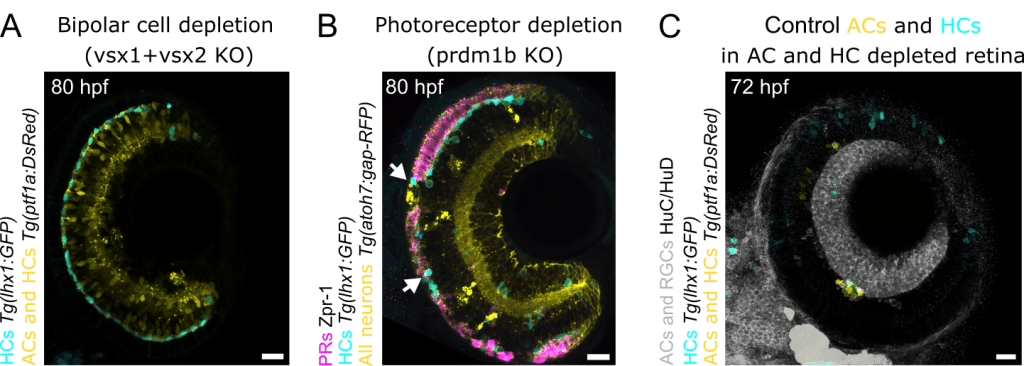
Could neighboring neurons provide contact-based or other forms of support for HC navigation? The retina consists only of five neuronal cell types (Figure 1A), which allowed us to systematically prevent each type from forming and observe how HCs would behave in these scenarios.
Previous postdoc from Norden lab had already removed retinal ganglion cells without observing a significant effect on HC migration (Amini et al., 2022). We therefore started by removing bipolar cells (as in Letelier et al., 2023) that occupy the retinal space through which HCs move. Unexpectedly, the layer normally filled by bipolar cells was now replaced by amacrine cells (AC), which expanded to occupy two retinal layers. Nevertheless, HCs still migrated successfully to their correct layer (Figure 2A).
Removing photoreceptor (PR) presented a challenge. As most prevalent and evolutionary ancient retinal neuronal type, they are notoriously difficult to deplete, shown by Elisa Nerli, previous PhD student in the lab (Nerli et al., 2023). After several F0 crispants trials, we eventually produced larvae with large patches lacking PRs. To our surprise, migrating HCs invaded these gaps, moving beyond their normal stopping point (Figure 2B). This suggested that, aside from acting as a physical barrier, PRs are not essential for HC migration.
The last neuron type ACs was particularly intriguing as HCs intermingle with them, prior to starting their multipolar migration. A well-established morpholino-based knockdown approach to remove AC existed, unfortunately also preventing HC generation. To examine HC migration in ACs absence, we therefore took an alternative strategy: transplanting control cells into embryos depleted of both HCs and ACs.
This produced a striking phenotype. Transplanted control HCs showed no upward movement towards the PRs and instead migrated only within the presumptive AC layer (Figure 2C). ACs thus appeared to be the key players in HC navigation towards their correct place of function.
Coordinated pushing and pulling keeps horizontal cells in line
The results suggested that ACs somehow promote HC migration whereas PRs physically restrict excess migration. This insight allowed us to focus our transcriptomics-based screen for potential ligand-receptor interactions on these three cell types.
After tedious dissection and processing of numerous retinae across developmental stages spanning HC migration, we obtained transcriptomic datasets predicting repulsive ligand-receptor interactions between ACs and HCs, as well as PR-derived attractive ligands potentially interpreted by HCs. Inspired by the feasibility of generating F0 knockout zebrafish larvae (Kroll et al., 2021), allowing results within about a week from setting crosses, I started screening candidate ligands and receptors with a simple readout: are the HCs in their correct layer after retinal layering is complete?
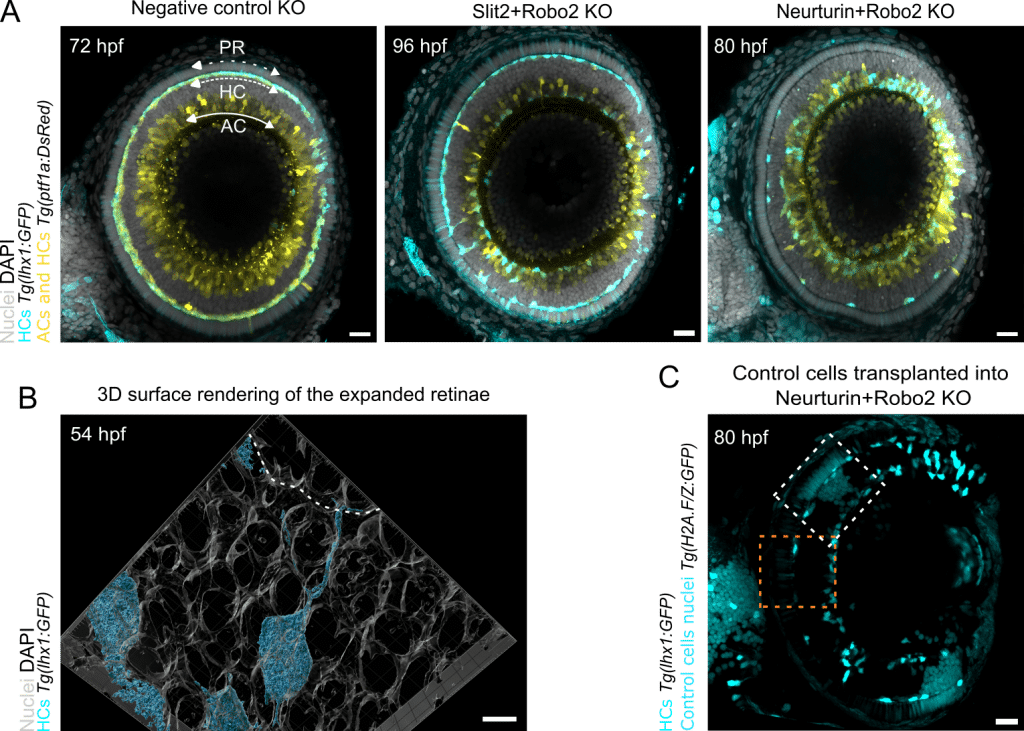
Removing Slit2 (repulsive ligand upregulated in ACs) or their Robo2 receptor (present in HC protrusions) produced an interesting phenotype. Roughly half of the HCs reached the PR layer while the other half remained with the ACs (Figure 3A). Live imaging further revealed that this behavior depended on the distance of HCs to the PR layer: the closer a HC was to the PRs, more likely it was to migrate successfully.
Without Slit-Robo signaling, HCs were no longer pushed away from ACs, toward the PR layer. Nevertheless, some HCs still managed to reach the PRs, suggesting that additional directional cues might be involved.
This led us to ask what else HCs might sense with their long protrusions as they extend in three dimensions within the crowded retinal tissue. To investigate this, we joined forces with Mario Del Rosario from Ricardo Henriques’ lab to perform expansion microscopy, physically expanding the zebrafish larvae four-fold.
Seeing enlarged retinae for the first time was truly impressive, although the working distance of our objectives quickly became the limiting factor. With optimized sample mounting, we could however observe beautiful HC protrusions weaving between neighboring cells to reach the PRs (Figure 3B). Perhaps photoreceptors express something HCs are attracted towards, explaining why some HCs still successfully migrate in the absence of Slit-Robo signaling?
Rewinding a bit, our transcriptomic analysis had also revealed the upregulation of the attractive ligand Neurturin in PRs, along with its receptors Gfrα1-2/Ret in HCs. We had also knocked these out individually, without any effect on HC migration. I was puzzled about this and Caren already wanted to push the paper out. Then a thought occurred to me: could Slit-Robo repulsion be so dominant that it masks weaker navigational cues?
To test this idea, I combined the Robo2 knockout (removing repulsion from ACs) with the removal of Neurturin or its Ret receptor. This turned into one of those rare ‘this is why I do science’ -moments at the Airyscan microscope. Now almost all HCs failed to migrate and instead remained stuck with the ACs (Figure 3A).
To further prove that PRs are the source of attractive Neurturin, Margarida Cruz, a PhD student in the Norden lab, performed a challenging transplantation experiment. She first removed both Robo2 and Neurturin in embryos expressing a HC reporter. Into these double-knockout embryos, she transplanted control cells with labelled nuclei, generating mosaic retinae containing patches of control PRs. As observed earlier for Neurturin+Robo2 knockout, most HCs failed to move. However, beneath the patches of control PRs we could now see HCs that had migrated successfully (Figure 3C).
To sum up, two different neuronal types on opposite sides of the retina coordinate push-pull mechanism to control HC multipolar migration: ACs begin to repel HCs upwards. Simultaneously, PRs secrete attractive Neurturin to ensure that HCs reach their final destination and form correct neuronal layer.
What’s next?
During brain development, differentiating neurons require precise directional cues to reach and stop in their correct layer. In the mammalian brain, multipolar migration often appears to be a transitional phase whereas in birds and reptiles it seems to represent a predominant migration mode used to build parts of the brain (Nomura et al., 2020; Scott et al., 2012).
In the zebrafish retina, we have also observed that all neurons except bipolar cells, can use multipolar migration as a backup strategy to reach their final location. Understanding how multipolar migration is directed in the retina should therefore provide insights that apply also to other parts of the developing brain.
The in vivo nature of our approach imposed certain limitations when trying to pinpoint the precise molecular interactions between neurons. Like Bilbo traveling there and back again, complementary experiments across scales are needed: in vitro approaches to probe molecular mechanisms of protrusion-mediated cell communication, ex vivo systems to examine tissue-level behavior and in vivo studies to observe how these interactions unfold within the developing organism. Having recently shifted from horizontal cells to investigate brain macrophages, microglia, which also move in a multipolar fashion, I am currently trying to apply these principles to understand their protrusion biology.
References
Amini, R., Bhatnagar, A., Schlüßler, R., Möllmert, S., Guck, J., & Norden, C. (2022). Amoeboid-like migration ensures correct horizontal cell layer formation in the developing vertebrate retina. eLife, 11, 1–30. https://doi.org/10.7554/elife.76408
Hahn, J., Monavarfeshani, A., Qiao, M., Kao, A. H., Kölsch, Y., Kumar, A., Kunze, V. P., Rasys, A. M., Richardson, R., Wekselblatt, J. B., Baier, H., Lucas, R. J., Li, W., Meister, M., Trachtenberg, J. T., Yan, W., Peng, Y.-R., Sanes, J. R., & Shekhar, K. (2023). Evolution of neuronal cell classes and types in the vertebrate retina. Nature, 624, 415. https://doi.org/10.1038/s41586-023-06638-9
Kroll, F., Powell, G. T., Ghosh, M., Gestri, G., Antinucci, P., Hearn, T. J., Tunbak, H., Lim, S., Dennis, H. W., Fernandez, J. M., Whitmore, D., Dreosti, E., Wilson, S. W., Hoffman, E. J., & Rihel, J. (2021). A simple and effective F0 knockout method for rapid screening of behaviour and other complex phenotypes. eLife, 10, e59683. https://doi.org/10.7554/eLife.59683
Lehtimäki, J. I., Lilue, J., Cruz, M. R., Rosario, M. D., Nerli, E., Henriques, R., & Norden, C. (2026). Spatiotemporal coordination of Slit-Robo repulsion and neurturin-Gfrα attraction guides multipolar migration during retinal lamination. Cell Reports, 45(2). https://doi.org/10.1016/j.celrep.2026.116948
Letelier, J., Buono, L., Almuedo-Castillo, M., Zang, J., Mounieres, C., González-Díaz, S., Polvillo, R., Sanabria-Reinoso, E., Corbacho, J., Sousa-Ortega, A., Corral, R. D. D., Neuhauss, S. C. F., & Martínez-Morales, J. R. (2023). Mutation of vsx genes in zebrafish highlights the robustness of the retinal specification network. eLife, 12. https://doi.org/10.7554/eLife.85594
Nerli, E., Kretzschmar, J., Bianucci, T., Rocha‐Martins, M., Zechner, C., & Norden, C. (2023). Deterministic and probabilistic fate decisions co‐exist in a single retinal lineage. The EMBO Journal, 42(14). https://doi.org/10.15252/embj.2022112657
Noctor, S. C., Martinez-Cerdeño, V., Ivic, L., & Kriegstein, A. R. (2004). Cortical neurons arise in symmetric and asymmetric division zones and migrate through specific phases. Nature Neuroscience, 7(2), 136–144. https://doi.org/10.1038/nn1172
Nomura, T., Ohtaka-Maruyama, C., Kiyonari, H., Gotoh, H., & Ono, K. (2020). Changes in Wnt-Dependent Neuronal Morphology Underlie the Anatomical Diversification of Neocortical Homologs in Amniotes. Cell Reports, 31(5). https://doi.org/10.1016/j.celrep.2020.107592
Ruoslahti, E. (1996). Brain extracellular matrix. Glycobiology, 6(5), 489–492. https://doi.org/10.1093/GLYCOB/6.5.489
Scott, B. B., Gardner, T., Ji, N., Fee, M. S., & Lois, C. (2012). Wandering Neuronal Migration in the Postnatal Vertebrate Forebrain. Journal of Neuroscience, 32(4), 1436–1446. https://doi.org/10.1523/JNEUROSCI.2145-11.2012
Tabata, H., & Nakajima, K. (2003). Multipolar Migration: The Third Mode of Radial Neuronal Migration in the Developing Cerebral Cortex. Journal of Neuroscience, 23(31), 9996–10001. https://doi.org/10.1523/jneurosci.23-31-09996.2003


 (No Ratings Yet)
(No Ratings Yet)