Imaging spotlight: Altair-LSFM
Posted by FocalPlane, on 9 March 2026
In this paper highlight from Kevin Dean and colleagues, we learn about Altair-LSFM, a light-sheet fluorescence microscope that they designed so it can be built and operated in biology labs without needing a full-time optical engineer.
Can you tell us why you developed Altair-LSFM and describe its key features?
Altair-LSFM grew out of a simple frustration that many microscopists share: the field moves fast, but commercialization is a slow process. Over the past ~50 years, advances in optics, cameras, probes, and computation have enabled extraordinary biology, yet it typically takes many years for a new imaging approach to become commercially available, and by the time it does, the community has often already moved on. Our goal with Altair-LSFM is to shorten that “idea-to-impact” gap by delivering a high-performance light-sheet microscope that researchers can actually build, operate, and adapt, without needing a full-time optical engineer. We focused first on a use case where this gap is especially costly: quantitative 4D imaging of sub-cellular dynamics in adherent cells.
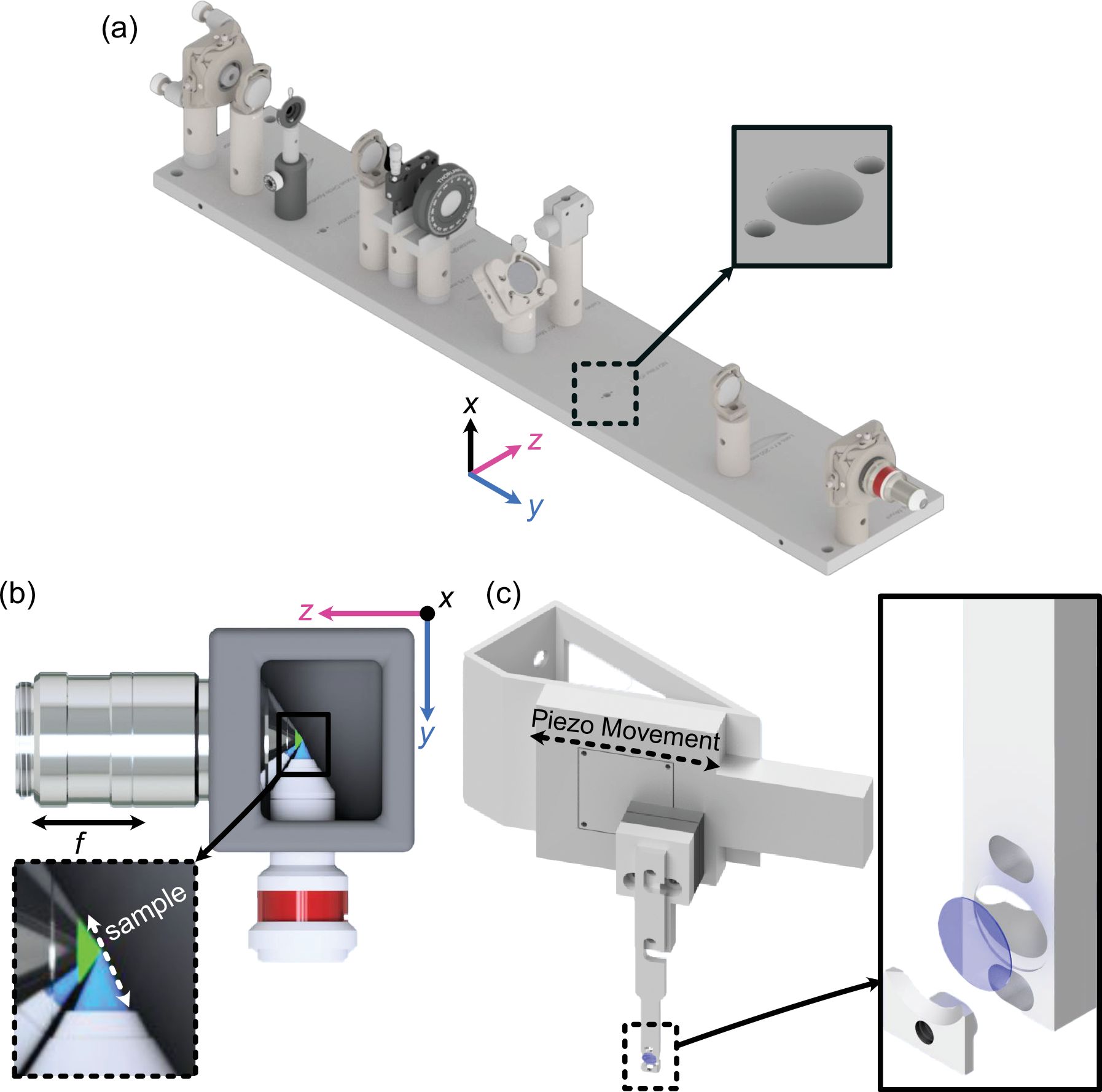
A big motivation for this specific system is that quantitative cell biology increasingly demands true Nyquist sampling in both space and time. If you want to measure dynamic three-dimensional processes such as cytoskeletal remodeling, organelle transport, or cell migration with a light-sheet microscope, you will need to acquire hundreds of image planes per volume per channel, rapidly, for minutes to hours. That pushes you into a regime where speed and sensitivity are everything: you either collect enough photons quickly (with low illumination burden), or you miss the biology.
Altair-LSFM is our first “buildable” iteration aimed squarely at that problem: sub-cellular, volumetric imaging in adherent cultured cells. Its key design choices reflect that focus:
- Maximized sensitivity and resolution through the detection path. We built the system around a high-NA, water-dipping detection objective, paired with a short, high-throughput detection train (minimal optical surfaces) and modern scientific CMOS cameras. This combination is what allows fast, wide-field readout while still collecting photons efficiently, critical for live imaging.
- Illumination designed for high axial resolution and gentle imaging. We use a tightly focused light sheet to sharpen optical sectioning and incorporate shadow-mitigation strategies (multi-directional beam pivoting) so structures don’t disappear behind scattering or absorption features. We also favor 100% duty-cycle illumination, which helps reduce peak intensities and supports gentler time-lapse imaging compared to approaches that rely on dithered or digitally scanned schemes.
- Performance that reaches the sub-cellular regime without exotic optics. The system achieves approximately 235 nm lateral and 350 nm axial resolution across a 266 µm field of view after deconvolution, performance that is competitive with the sub-cellular end of light-sheet imaging while staying grounded in a reproducible, buildable design.
- Designed for dissemination, not just a single lab. Many open-source light-sheet systems are fantastic but are optimized for embryos, large, cleared tissues, or lower magnification regimes. Altair-LSFM was designed specifically to fill the open gap for sub-cellular imaging, and we put a lot of emphasis on reducing assembly and alignment pain. The optical system is translated from in-silico optimization into precision baseplate hardware, which removes unnecessary alignment degrees of freedom and makes assembly far more approachable for non-experts.
- Software that lowers the barrier even further. Altair-LSFM is operated through our open-source control platform, navigate, which is built to be high-performance and “smart microscopy” ready, without requiring end users to write code.
In short, Altair-LSFM is meant to be a compact, easy-to-assemble light-sheet microscope that excels at imaging adherent cells, delivering sub-cellular resolution, multicolor capability, and a practical workflow at a price point (~$150k fully equipped, including optical table and computer workstation assemblies) that is substantially lower than typical commercial high-resolution light-sheet systems. The long-term vision is a modular family of microscopes that biologists can deploy quickly, use reliably, and evolve with the field, so the instrumentation stops being the bottleneck and starts being the accelerator.
What are the prospects for further development?
So far, Altair-LSFM has served as both a validated sub-cellular imaging platform and a design template for what we want the next generation of “buildable” light-sheet systems to look like. In the paper, we used it for high-resolution fixed-cell imaging and live-cell demonstrations; since then, our primary focus has been using the same engineering philosophy (in-silico optimization → baseplate translation → turnkey operation) to rapidly build the next instruments in the Altair family.
The most immediate extension is Altair-ASLM, which places Axially Swept Light-Sheet Microscopy on a single baseplate and supports both aqueous and chemically cleared specimens. It includes an optimized, aberration-free remote focusing unit that provides deterministic scanning of the beam along its propagation direction, and improved illumination optics for even more uniform, high-resolution imaging. The aqueous implementation (using the same high-NA water-dipping lenses as Altair-LSFM) achieves ~235 nm lateral and ~350 nm axial resolution after deconvolution across a ~266 × 266 µm field of view. When paired with NA 0.7 multi-immersion objectives, we expect <300 nm isotropic resolution across approximately ~300 × 300 µm (testing ongoing).
In parallel, we’re extending Altair into oblique plane microscopy (OPM). Our first OPM iteration is a macroscale variant intended for large-area imaging, providing ~2 × 3 µm lateral resolution and ~15 µm axial resolution across an ~8 mm field of view—useful for rapid screening and multiscale workflows.
Finally, we’ve continued to push “non-expert operability” by simplifying the control stack. We have now eliminated the need for National Instruments DAQ hardware, consolidating stage control, filter wheels, and all analog/digital timing into a single commercially available controller that can be purchased pre-configured. Taken together, these developments reflect where we see Altair heading: a modular family of baseplate-mounted light-sheet microscopes, spanning sub-cellular to macroscale imaging and aqueous to cleared specimens, all designed to be deployable outside of specialist optics labs.
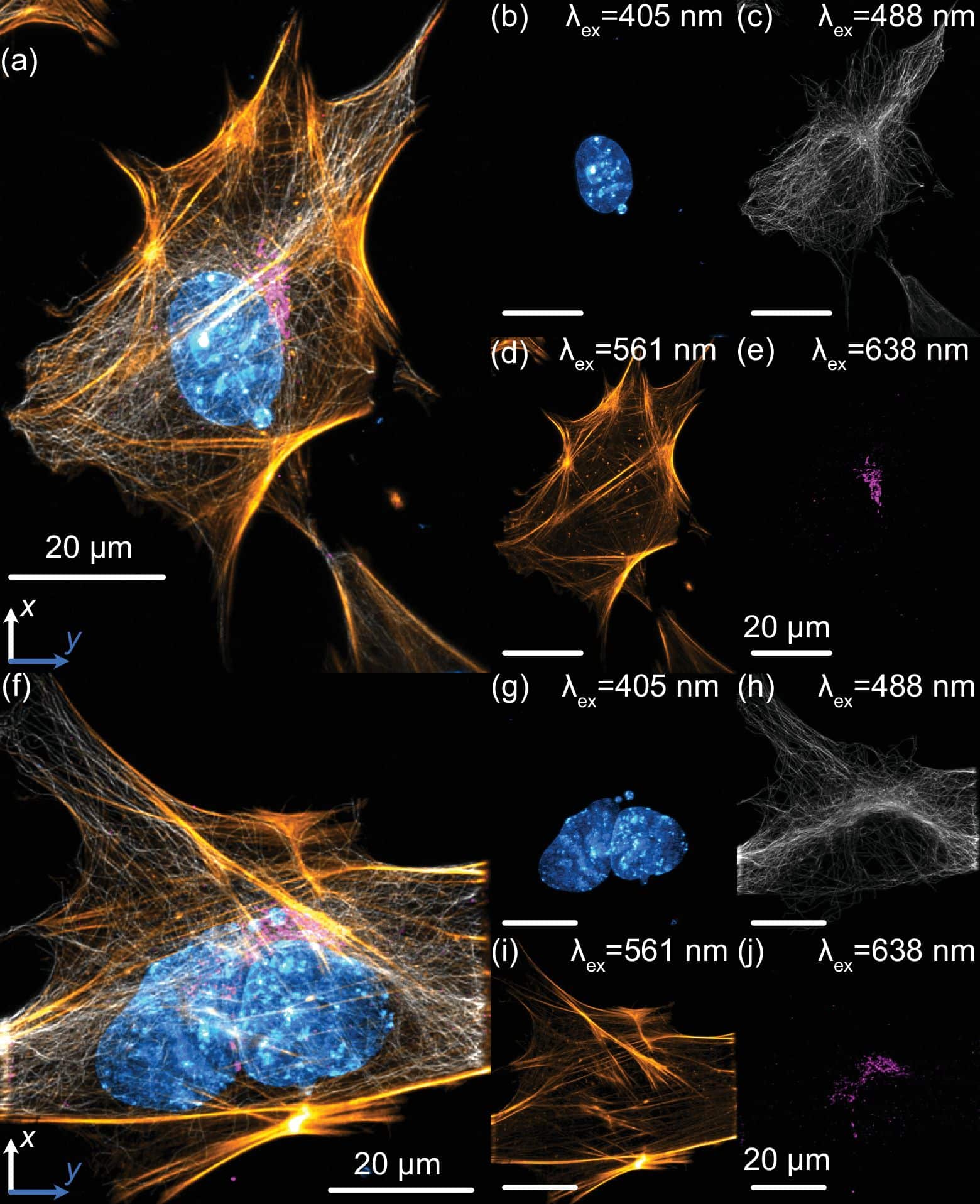
Do you have any tips or tricks for researchers setting up or imaging with Altair-LSFM?
Absolutely—here are a few practical tips that have helped us (and that we hope will help new builders) get up and running smoothly with Altair-LSFM:
- Start with the documentation and follow it in order. We put a lot of effort into making the build process reproducible and approachable. The most reliable path is to use the step-by-step guide at: thedeanlab.github.io/altair
- Use GitHub Issues like a help desk (seriously). Don’t be shy—if anything is unclear, missing, or doesn’t match what you’re seeing on your bench, please open an issue here: https://github.com/TheDeanLab/altair/issues. Even small questions help. We treat the repository as a living resource, and real user feedback is what makes it better over time.
- Validate in layers: beam → sheet → beads → cells. A simple workflow that prevents “mystery problems” later is:
a) confirm the beam is collimated and centered through the optical train
b) verify the light sheet looks right (thickness + uniformity)
c) measure PSF with beads
d) then move to cells
Catching issues early saves a lot of time later.
- Be intentional with alignment degrees of freedom. Altair-LSFM is intentionally designed to eliminate most alignment knobs, so stick to the intended adjustments (laser collimation, galvo rotation, folding mirror tip/tilt, and objective positioning). If the beam doesn’t look right, stop and identify the root cause immediately, it won’t magically improve later in the optical train. The most reliable way to verify alignment is to use back-reflections from optical surfaces and strategically placed irises, both of which are outlined step-by-step in our documentation.
- For imaging: prioritize photon efficiency over laser power. Especially for live-cell imaging, resist the temptation to crank up illumination. I strongly prefer keeping peak laser intensity as low as possible; if more signal is needed, I would rather increase exposure time (or modestly average) than increase laser power. Photobleaching and phototoxicity are often nonlinear with intensity, so a 2× increase in illumination can produce more than a 2× increase in photodamage, even when the total delivered dose seems comparable.
If you build Altair-LSFM (or even start planning one), we’d love to hear from you; your questions and comments will directly shape the next iteration of the docs and hardware.
Where can people find more information?
You can find the full technical details in our manuscript and the complete build/assembly documentation, including CAD, parts lists, and protocols, at thedeanlab.github.io/altair (with issues/support via the GitHub repo).


 (No Ratings Yet)
(No Ratings Yet)