Imaging spotlight: the axonal membrane-associated periodic skeleton
Posted by FocalPlane, on 5 March 2026
In this paper highlight, Nick Boyer takes us through the key results and imaging techniques that they used in their research dissecting the axonal membrane-associated periodic skeleton with Rohan Sharma, Christophe Leterrier, Subhojit Roy and colleagues.
What are the key results from your paper?
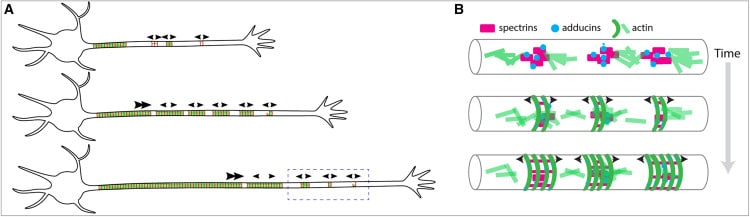
In this study we examined the development of the axonal membrane-associated periodic skeleton (MPS), an exceptional structure which confers stability to the neuronal axon and acts as a signaling scaffold. The fully-developed MPS is highly ordered, consisting of circumferential rings of actin every ~180 nm spaced by spectrins, and spans hundreds to thousands of microns of the axon. In juxtaposition to this incredible degree of structure, our study showed that the nascent MPS is composed of disordered patches of component proteins. Using STORM, SIM, and confocal microscopy and fluorescence recovery after photobleaching (FRAP) analysis we found that these patches are consistent with liquid-like biomolecular condensates. We performed structure-function FRAP assays on condensates formed by partial constructs of spectrin proteins and determined that the coiled-coil rich spectrin repeat domains likely drive this behavior. Using these findings we constructed a new model for how the MPS develops in which components are concentrated in biomolecular condensates, and then subsequently assemble into the mature axon-spanning structure.
What imaging techniques have you used in your research?
While we employed a variety of imaging techniques, we relied most heavily on STORM and SIM super-resolution microscopy and confocal FRAP assays. The beautiful STORM imaging by the lab of Christophe Leterrier gave us critical insight into the structure of patches of spectrins in distal axons. Though these patches were large enough and separated enough to be resolved by conventional microscopy, super-resolution allowed us to see that only some patches more proximal to the soma contained the expected lattice structure of mature MPS, while more distal patches displayed either partial lattice or a complete lack of structure. This conventional versus super-resolution divide mirrors the discovery of the MPS itself, whose ~180nm repeat period hid it from prying eyes until the advent of STORM imaging. The results of the Leterrier lab’s imaging, which showed a surprising lack of structure, led us to employ FRAP analysis to probe the internal structure of these disorganized spectrin patches.
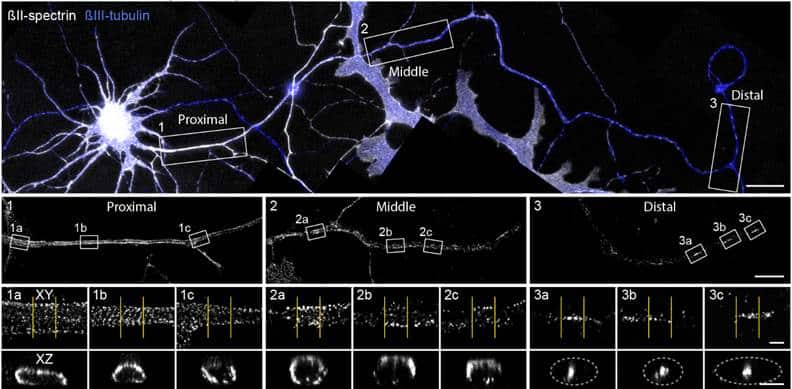
We first bleached whole spectrin patches in distal axons and observed the recovery on a widefield fluorescence microscope. The large and deep field of view allowed us to quickly find multiple neurons with only a low expression level of transfected αII-spectrin:GFP. Low expression was critical as high expression of spectrins has been shown previously to produce a mostly soluble population rather than allowing incorporation of the spectrin into native structures. Fluorescence of isolated patches slowly recovered suggesting more loosely bound spectrin, in contrast to a lack of fluorescence recovery in the proximal axon where spectrin lattice assembly was expected to be complete. When expressed in HEK293T cells spectrins produced inclusions which similarly slowly recovered fluorescence after photobleaching.
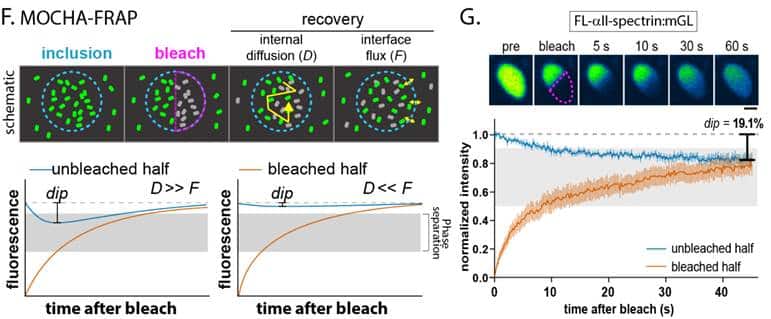
To further probe the internal structure of these spectrin inclusions we quantified the redistribution of fluorescence within them after photobleaching a portion of each structure using MOCHA-FRAP analysis. The balance between diffusion within inclusions and exchange of protein with surrounding cytoplasm can delineate between liquid droplets and other forms of biomolecular condensation. Using MOCHA-FRAP we found that the properties of αII- and βII-spectrin droplets were consistent with liquid-liquid phase separation, while inclusions of other MPS proteins like adducin were more solid-like. Given the challenges of purifying extremely large proteins like spectrins for traditional phase separation assays, an imaging technique like MOCHA-FRAP proved to be the ideal companion to super-resolution microscopy to determine that liquid-like spectrin condensation is an early stage in the development of the MPS.
Are there technical tips or tricks that you have learnt doing this research?
To perform FRAP assays in distal axons we needed fluorescently-tagged spectrins properly distributed in the cell, with low enough protein levels to avoid overexpression artifacts. Generating a stable mouse line with a fluorescent tag would have been prohibitively expensive and time consuming for the small set of planned assays, and endogenous tagging proved too slow of an onset to study early development of the relatively long-lived spectrin proteins. We therefore turned to plasmid lipofection with some modification to the manufacturer protocol to accommodate quirks of the spectrins. To account for the size of the tagged spectrin plasmids (>11,000 bp) we found that doubling the maximum recommended DNA per transfection for spectrins produced the best results. We coupled this with a 20% reduction from the lowest manufacturer recommended concentration of lipofection reagent to reduce toxicity to our fragile developing neurons. The critical modification was the timing of transfection and imaging. Due to the sensitivity of sparsely cultured, developing neurons, we replaced transfection media with a 50:50 mixture of fresh and conditioned culture media after 4 hours of incubation. We then waited at least 40 hours between transfection and imaging to allow newly translated, tagged spectrin to be ferried to the distal axon by slow axonal transport. These adjustments to the standard transfection protocol allowed us to successfully label spectrin patches without evident alterations of their structure.
Are there any advances in imaging or image analysis that would help your research?
FRAP assays are increasingly popular tools to measure physical properties of intracellular structures, especially liquid-like condensates. Despite this, a number of flaws with the most commonly used fluorophores make precise measurements of rapid processes, like measuring diffusion of very small proteins in condensates, difficult if not impossible. The fluorophores used for many FRAP studies remain primarily proteins like GFP or GreenLantern, as they can be genetically encoded and are highly susceptible to photobleaching. However, destruction of the fluorophore requires high intensity light for long periods of time relative to the scale of intracellular dynamics. Additionally, reactive oxygen species produced by photobleaching can damage the cell and alter the very processes FRAP is intended to measure.
To this end, one of the most important advances for FRAP would be development of new fluorophores with ideal bleaching properties. The bleaching of the new fluorophore would need to remain spatially precise and be as rapid as possible, with minimized input energy to reduce unintended damage. At the same time the fluorophore would need to be relatively photostable under normal imaging conditions with good quantum yield. It would need to be either fully genetically encodable or make use of adaptors like Halotag or SNAP-tag as this is key to many FRAP applications, especially with the increasing usage of endogenous protein tagging Developing a probe with all of these qualities is no small task, hence the dominance of GFP and related proteins for so long. However, every step towards the ideal would be a step in the right direction and would improve FRAP assays along the way.


 (No Ratings Yet)
(No Ratings Yet)